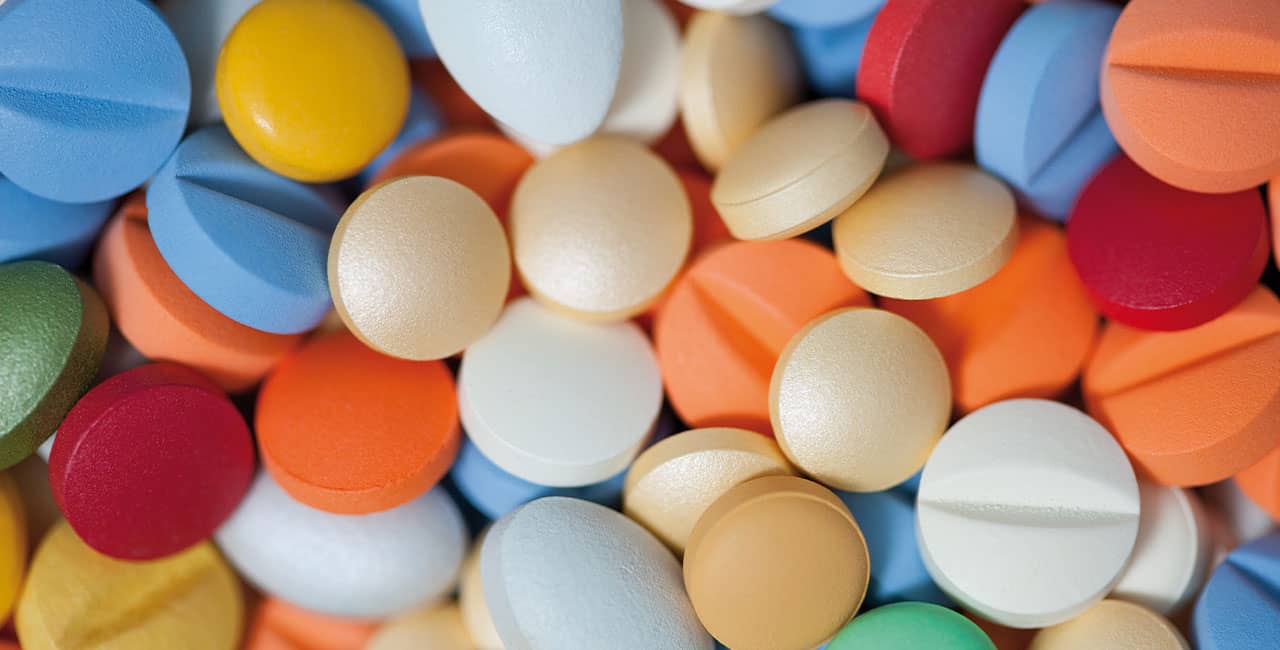
You are looking for quality?
Challenge our knowledge
Discover our product variety!
TITANIUM DIOXIDE FREE – discover our AquaPolish® film coating systems. Formulated without TiO2. For brilliant white and colored tablet surfaces. Our formulations achieve high opacity and offer a wide variety of natural color options.
AQUAPOLISH® PRO – the immediate release film coating provides a moisture barrier effect for solid oral dosage forms. In addition, it is a highly flexible film with good taste masking properties. For a smooth tablet surface.
INGREDIENT PREMIXES free of undesired excipients like titanium dioxide, silicon dioxide, talc, magnesium stearate, stearic acid and synthetic colors. For clean label film coating and tableting of solid oral dosage forms.
Excellence still remains
The range of applications for BIOGRUND products is still growing. Our focus is now no longer just on film coating systems. This fact has prompted us to adapt the design of our company logo. In order to be able to give more attention to the entire product portfolio, we decided to drop the logo addition “Filmcoating Excellence” for all BIOGRUND companies. Meanwhile, the expanded portfolio includes excellent ready-to-use premixes for tableting and coloring, which are complemented by a range of excellent excipients such as celluloses, waxes and cleaners.
For the transition period, customers may encounter both the old logo with the subline “Filmcoating Excellence” and the new logo without this subline on packaging material, labels or documents. Here it should be clear that it is one and the same company.